TaBriX is developing innovative solutions to create anti-infectives for the future.
This project focuses on the development of drugs that target novel, unexplored biological pathways to treat Tuberculosis and tackle the global threat of drug resistance.

Founder of TaBriX, Prof. Lydia Tabernero
Tuberculosis (TB) is a major health problem, which affects approximately 25% of the human population and kills 1.6m people per year. One of the main obstacles in the cure and eradication of TB is antibiotic resistance. There are roughly half a million new cases of drug-resistant TB per year, with an alarming increase of extensive or untreatable drug-resistant cases.
The current antibiotic treatments for TB are long and complex and do not completely clear the infection, which leads to relapse and drug resistance. Multi Drug Resistant (MDR) and Extensively Drug Resistant (XDR) TB treatments are longer, more toxic and less effective. Current drugs are incompatible with HIV therapy, thus contributing to comorbidity and transmission.
To cure and eradicate TB, the most significant hurdle to overcome is antibiotic resistance. Traditional anti-TB therapeutics act by inhibiting essential bacterial processes that result in drug resistant Mycobacterium tuberculosis (Mtb) strains (the bacteria that causes TB).
There is a need for shorter, simpler, and more effective treatments with new modes of action.

Prof. Lydia Tabernero examines the structure
An alternative approach is to inhibit Mtb virulence factors, thus compromising infection persistence. Mtb promotes its own survival by secreting virulence factors, which block the natural immune response and prevent host macrophages (the white blood cells that Mtb hide in) being able to kill the bacteria.
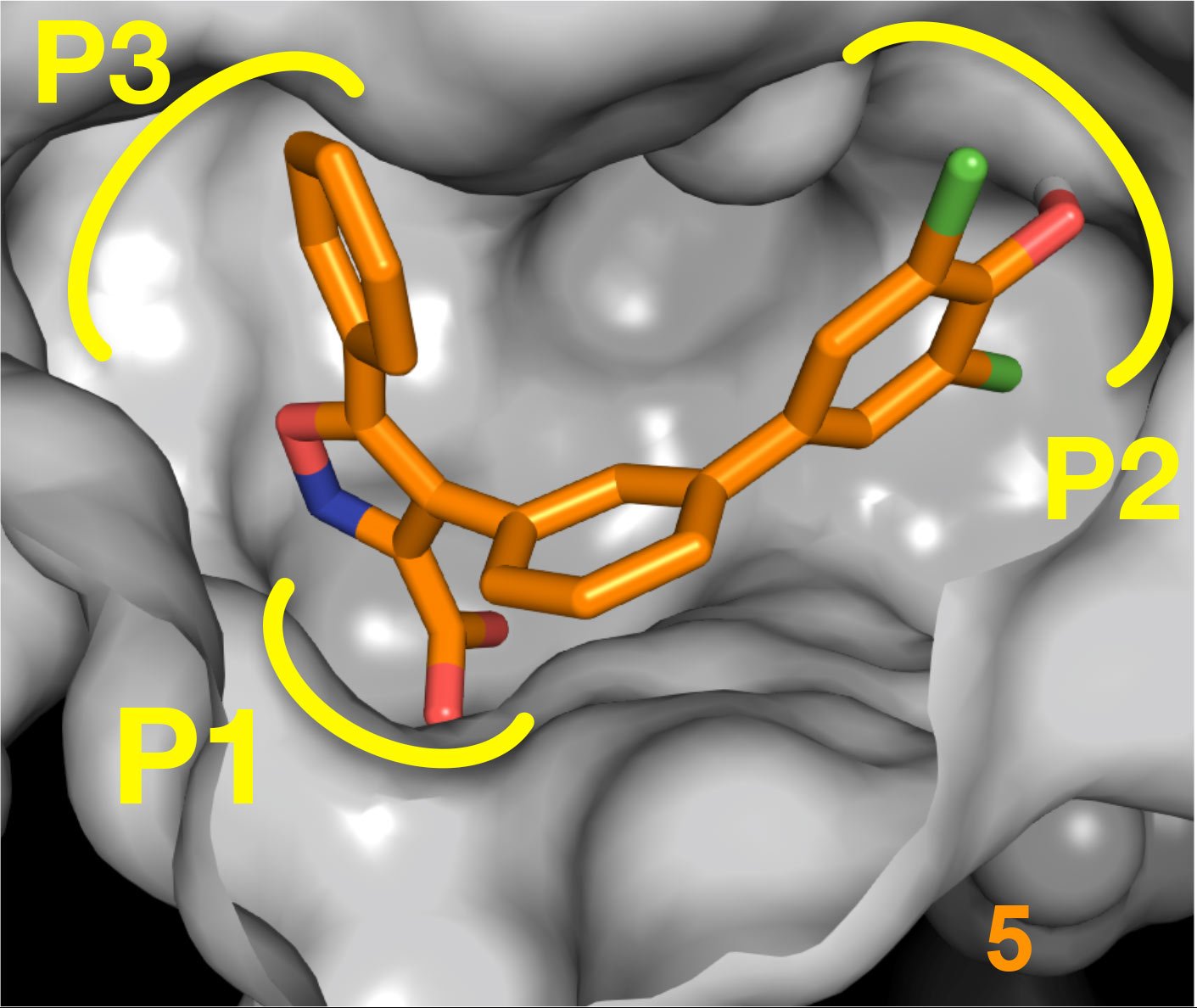
Professor Lydia Tabernero and her team have developed potent small-molecule inhibitors that target critical virulence factors and represent a novel therapeutic mode of action to treat TB. This project has the potential to tackle the global threat of drug resistant TB, and wider antimicrobial resistance.

Prof. Lydia Tabernero in the lab